In the vast landscape of healthcare, the term DMEPOS may sound like an acronym from a complex lexicon. However, understanding DMEPOS is crucial, especially for those involved in providing durable medical equipment, prosthetics, orthotics, and supplies. In this blog post, we’ll demystify the acronym, explore its significance, and delve into various aspects, including DMEPOS accreditation, Medicare DMEPOS application, DMEPOS bonds, and the intricacies of competitive bidding in this domain.
Decoding DMEPOS
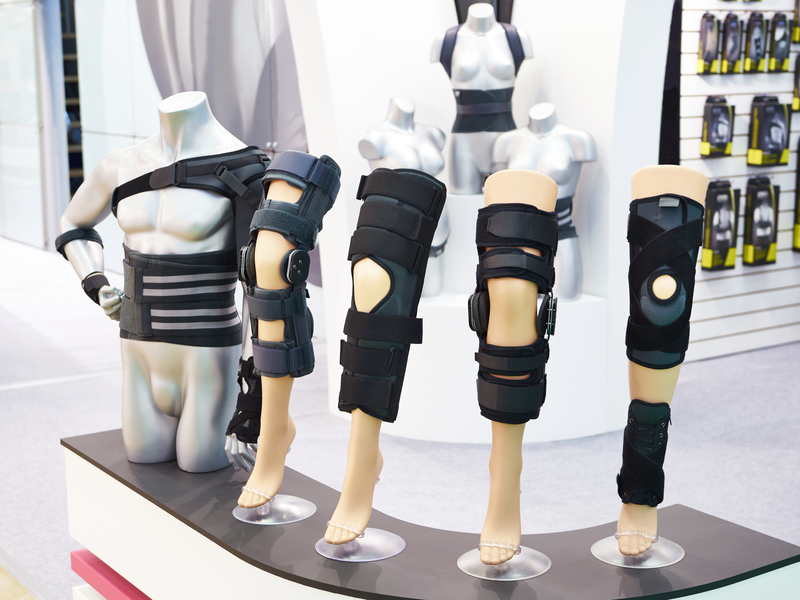
DMEPOS stands for Durable Medical Equipment, Prosthetics, Orthotics, and Supplies. It encompasses a broad category of healthcare items that serve a medical purpose, are durable, and are used repeatedly. This includes everything from wheelchairs, oxygen equipment, and diabetic supplies to orthotic devices and prosthetic limbs.
DMEPOS Accreditation
Providers of DMEPOS must adhere to strict quality standards to ensure the safety and well-being of patients. DMEPOS accreditation is a process by which healthcare suppliers are evaluated against established criteria to ensure compliance with Medicare standards. Accreditation is often required for suppliers to participate in Medicare, and it is an essential step to demonstrate a commitment to quality and patient care.
Medicare DMEPOS Application
For suppliers seeking to furnish DMEPOS items and services to Medicare beneficiaries, obtaining Medicare accreditation is a requisite. The Medicare DMEPOS application involves a thorough assessment of the supplier’s business operations, quality control measures, and adherence to applicable laws and regulations. Successful accreditation is a prerequisite for suppliers to bill Medicare for covered items and services.
DMEPOS Bonds
DMEPOS bonds, also known as surety bonds, are a financial requirement imposed by the Centers for Medicare & Medicaid Services (CMS) as part of the accreditation process. These bonds serve as a form of protection for Medicare and the beneficiaries. They ensure that suppliers comply with Medicare regulations, and in the event of non-compliance or fraud, the bond can be utilized to cover financial losses.
Competitive Bidding in DMEPOS
To control costs and improve the efficiency of DMEPOS procurement, Medicare has implemented a competitive bidding program. Under this program, suppliers submit bids to provide certain items in specific geographic areas. The competitive bidding process aims to secure quality items at lower costs for Medicare beneficiaries. It encourages suppliers to offer competitive prices while maintaining high standards of service and quality.
What Is DMEPOS: A Comprehensive Overview
At its core, DMEPOS represents a vital component of patient care, ensuring that individuals have access to durable medical equipment, prosthetics, orthotics, and supplies essential for their well-being. The comprehensive nature of DMEPOS encompasses a wide array of items that contribute to the comfort, mobility, and quality of life for patients facing various health challenges.
Conclusion
In navigating the intricate world of healthcare, understanding DMEPOS is pivotal for both providers and patients. DMEPOS accreditation, the Medicare DMEPOS application process, DMEPOS bonds, and the dynamics of competitive bidding collectively shape the landscape in which suppliers operate and patients receive critical healthcare items.
For suppliers, compliance with accreditation requirements and participation in Medicare programs not only opens doors to reimbursement but also underscores a commitment to delivering quality care. For patients, awareness of the significance of DMEPOS ensures access to reliable and high-quality medical equipment and supplies that can significantly impact their overall well-being.
As the healthcare industry continues to evolve, the role of DMEPOS remains integral in enhancing the quality of life for individuals with diverse medical needs. In embracing the principles and standards associated with DMEPOS, healthcare providers contribute to a system that prioritizes patient comfort, mobility, and overall health.



 By:
By: 
 By:
By: 
 By:
By: 
 By:
By: